
Then, the next electron leaves the 3d orbital and the configuration becomes: Ar4s 0 3d 6. The first two to go are from the 4s orbital and Cobalt becomes:Ar4s 0 3d 7. 8 Cobalt has a relative permeability two-thirds that of iron. 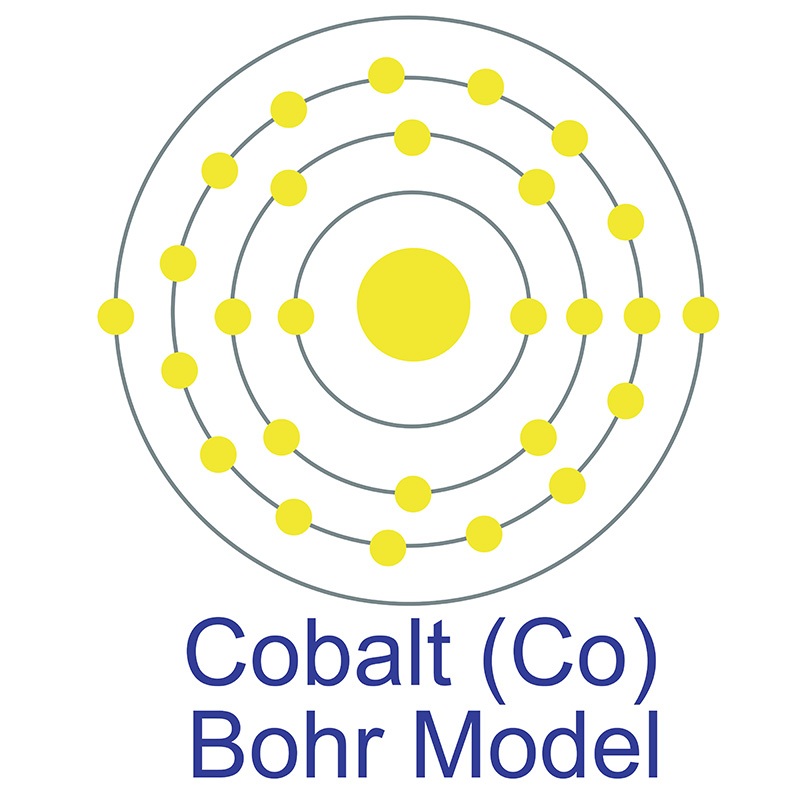
The Curie temperature is 1,115 C (2,039 F) 7 and the magnetic moment is 1.61.7 Bohr magnetons per atom. To save room, the configurations are in noble gas shorthand. Characteristics A block of electrolytically refined cobalt (99.9 purity) cut from a large plate Cobalt is a ferromagnetic metal with a specific gravity of 8.9. 
The electron configuration of Cobalt will end in d block as Cobalt is an inner transition metal. The electron configuration for Cobalt at ground state would simply be Co: Ar 4s23d7. positive electrical charges of the atomic nuclei expressed in electronic units. The s,p,d,f configuration for cobalt (Co) is 1s22s22p63s23p64s23d7, determined by the position of the element on the periodic table. This list of electron configurations of elements contains all the elements in increasing order of atomic number. The electron configuration of Cobalt is: 1s2 2s2 2p6 3s2 3p6 3d7 4s2. periodic table, in full periodic table of the elements, in chemistry. When observing Cobalt 3+, we know that Cobalt must lose three electrons. The electron configuration shows the distribution of electrons into subshells. Atomic no.Trending Questions What is the name of the moons changing shapes? The nearest star to Earth is Proxima Centauri which is 4.3 light-years away If light leaves this star in 2020 when will it reach Earth? Can latitude affect the angle of which the sun's rays hit earth? What is the speed of Halley's Comet when it is nearest Earth? What is the speed of fall on Earth vs on Moon? Why is reflectivity of insolation at high latitudes greater in winter than in summer? What is a sunspot and the pattern that exists in the number of sunspots that are observed? What characteristic do stars in a constellation or asterism share? How far away is NGC 5866? What is the source of all elements in the universe that are more massive than iron? Where does astronauts get water? How do you get into a parallel universe? While watching a star you see it moves 15 degrees across the sky how long have you been watching it? What scientists played a major role in overturning the ancient idea of an earth centered universe and when? How are low tides formed? How long would it take by spaceship to reach the vega star? Which is better observed by an X-ray telescope than an infrared telescope? This sphere is drastically tilted at 82. The electron configuration of Cobalt is Ar4s 2 3d 7. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. In order to write the electron configuration we first need to know the number of electrons for the Cobalt (Co) atom. Determine the number of core and valence electrons in an atom of cobalt. What is the short-hand electron configuration of cobalt(II), Co²+ ion Enter the electron configuration using no spaces or superscripts using the proper case letters (e.g., He 2s² 2p4 would be entered as He2s22p4). Write the condensed electron configuration of a cobalt, Co, atom (Z 27) using s, p, d notation. Electron configuration chart of all Elements is mentioned in the table below. Write the full electron configuration of a cobalt, Co, atom (Z 27) using s, p, d notation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
